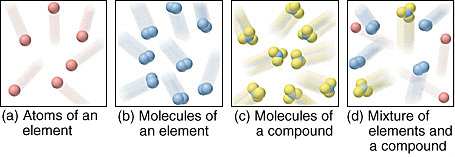
DIFFERENCE BETWEEN ELEMENTS, COMPOUNDS AND MIXTURES
Elements:
According to the Oxford Student's Science Dictionary (p. 8) an element is: a substance that cannot be broken down into two or more simpler substances by chemical (or physical) means.
The Teaching Primary Science textbook also explains that an element is a pure substance consisting of one type of atom (and all of the same size).
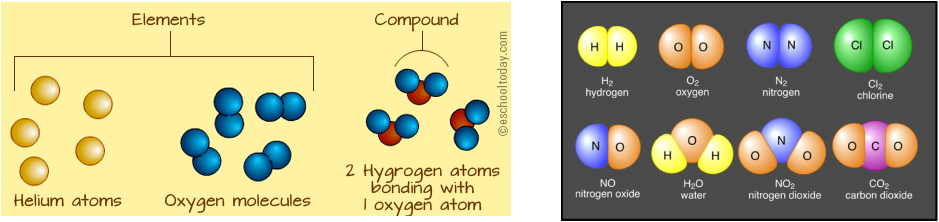
Examples of elements include: oxygen, carbon, zinc, copper, mercury and lithium (in batteries)
Note that an element:
- * consists of only one kind of atom
- * cannot be broken down into a simpler type of matter by either physical or chemical means, and
- can exist as either atoms (Image 'a' above) or molecules (image 'b' above)
- * the way the atoms are bonded together (molecular structure) doesn't matter
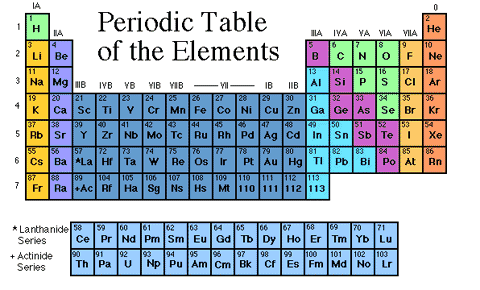
- * you can identify an element by its name because it will be listed on the periodic table (see below)
Compounds:
According to the Oxford Student's Science Dictionary, a compound is the substance formed by a chemical combination of elements in fixed proportions, as represented by the compound's chemical formula.
The creation of a compound from two more elements is described as a chemical or permanent change according the the Teaching Primary Science textbook.
Examples of compounds are: water, salt (sodium chloride), hydrogen peroxide and sugar.
Note that a compound:
* requires a chemical change to occur to happen
* every molecule is exactly the same size as every other molecule (see 'c' at top of page)
* when compounds form, a constant ratio of elements are made (see right image below)
* can only be separate into a simpler type of matter (elements) by chemical means, not physical
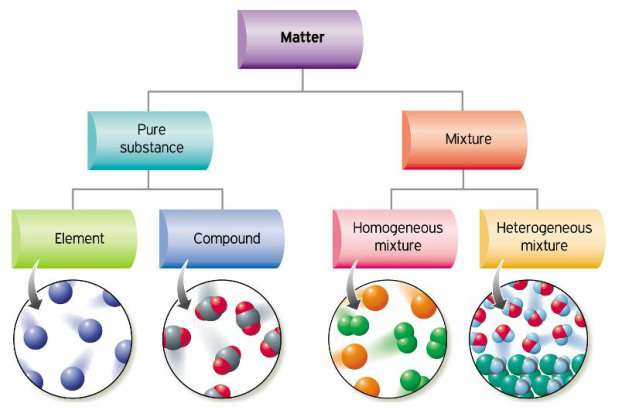
Mixtures:
A mixture is a combination of two or more substances that have not reacted chemically and can be separated using physical processes such as dissolving, crystallization, evaporation, etc.
Examples of mixtures are: milk, air, wood and salt water
Note that mixtures:
* are not pure substances
* are the most common form of organisation
* there are at least 2 different types of atom or molecule that are not chemically
combined or bonded to each other
* each item retains its properties in the mixture
Here's a fun online test to see what you know about compounds and mixtures: