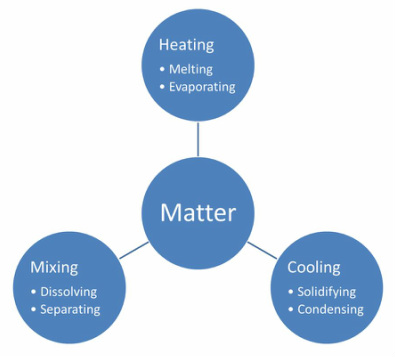
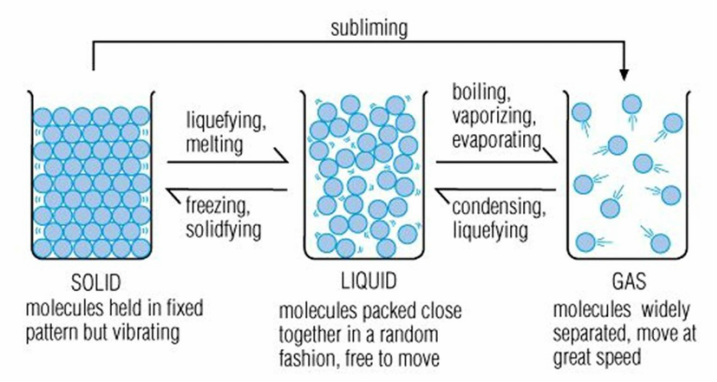
Processes involved in the changing states of matter
According to Loxley, Dawes, Nicholls, & Dore (2014, p.g. 276-277)
"Many substances are capable of changing from one form to another when they are either heated or cooled. The amount of heating or cooling needed to produce a change of state differs for each material. Changes of state are reversible or physical changes.
These processes can be explained by imagining materials are made up ultimately of tiny invisible particles that are held together by mutual attraction. This is the particle model. When a solid is heated, the particle gain energy and start to move away from each other and as they do so the solid becomes a liquid. As a liquid gains energy, the particle are able to break away from each other and become a gas. The reverse happens when material is cooled. As they gain or lose energy (heating or cooling), the physical properties of the material itself change."
"Many substances are capable of changing from one form to another when they are either heated or cooled. The amount of heating or cooling needed to produce a change of state differs for each material. Changes of state are reversible or physical changes.
These processes can be explained by imagining materials are made up ultimately of tiny invisible particles that are held together by mutual attraction. This is the particle model. When a solid is heated, the particle gain energy and start to move away from each other and as they do so the solid becomes a liquid. As a liquid gains energy, the particle are able to break away from each other and become a gas. The reverse happens when material is cooled. As they gain or lose energy (heating or cooling), the physical properties of the material itself change."
Examples of Change
If we look at a block of ice as an example:
As the ice, a solid, is exposed to more energy (heat) the particles gain more energy until they move more freely as they have broken the attraction between themselves that holds them together in a compacted form, the ice is now water - a liquid. As the energy levels continue to gain momentum (more heat) and eventually break free of each other. The water is now vapour - a gas.
As the ice, a solid, is exposed to more energy (heat) the particles gain more energy until they move more freely as they have broken the attraction between themselves that holds them together in a compacted form, the ice is now water - a liquid. As the energy levels continue to gain momentum (more heat) and eventually break free of each other. The water is now vapour - a gas.
Exceptions to the change process
There are some mixtures or solutions that require more than the simple steps outlined above for the matter to change its state. Solutions are a combination of matter types and require separation through a number of different ways.
For example:-
Sieving is required to separate a combination of sand and water.
Settling and pouring is needed to separate liquids such as vinegar and oil.
Filtering would be used to separate oxygen and dust particles.
Evaporation is required to separate elements such as salt and water.
These are all referred to as mechanical processes.
There are some elements that wen combined require more than a one step process to be separated. For example a combination of salt and sand would need an additional element added in order to separate the original two elements. Water would have to be added to the mixture to create a new solution. This solution could be sieved to remove the solid particles of sand. Then, the remaining solution would be heated to use evaporation to separate the remaining salt and water.
For example:-
Sieving is required to separate a combination of sand and water.
Settling and pouring is needed to separate liquids such as vinegar and oil.
Filtering would be used to separate oxygen and dust particles.
Evaporation is required to separate elements such as salt and water.
These are all referred to as mechanical processes.
There are some elements that wen combined require more than a one step process to be separated. For example a combination of salt and sand would need an additional element added in order to separate the original two elements. Water would have to be added to the mixture to create a new solution. This solution could be sieved to remove the solid particles of sand. Then, the remaining solution would be heated to use evaporation to separate the remaining salt and water.